|
We demonstrated the performance of the system through various experiments, including imaging standard targets, utilizing an eye phantom, and conducting in vivo imaging of the human retina. Once trained, our algorithm can reconstruct a megapixel CASSI image with only 60 s, a 20 times improvement compared with conventional iterative algorithms. Moreover, to enable fast and high-quality image reconstruction, we developed a deep-learning-based algorithm. The average spectral resolution is 5 nm from 445 nm to 602 nm. The resultant system can capture a 1180 × 1100 × 35 (x, y, λ) spectral datacube in a snapshot with a 17.5 μm and 15.6 μm resolution along the horizontal and vertical axes, respectively. In this work, we developed a spectral imaging device based on CASSI and integrated it with a commercial fundus camera. Therefore, the acquisition efficiency of CASSI is significantly higher than non-compressive methods that directly map spectral datacube voxels to camera pixels, such as IMS. Essentially, just one spectrally dispersed projection of the datacube that is spatially modulated by the aperture code over all wavelengths is sufficient to reconstruct the entire spectral datacube 14. Provided that the spectral datacube is sparse in a given domain, it can faithfully be reconstructed with a regularization method. 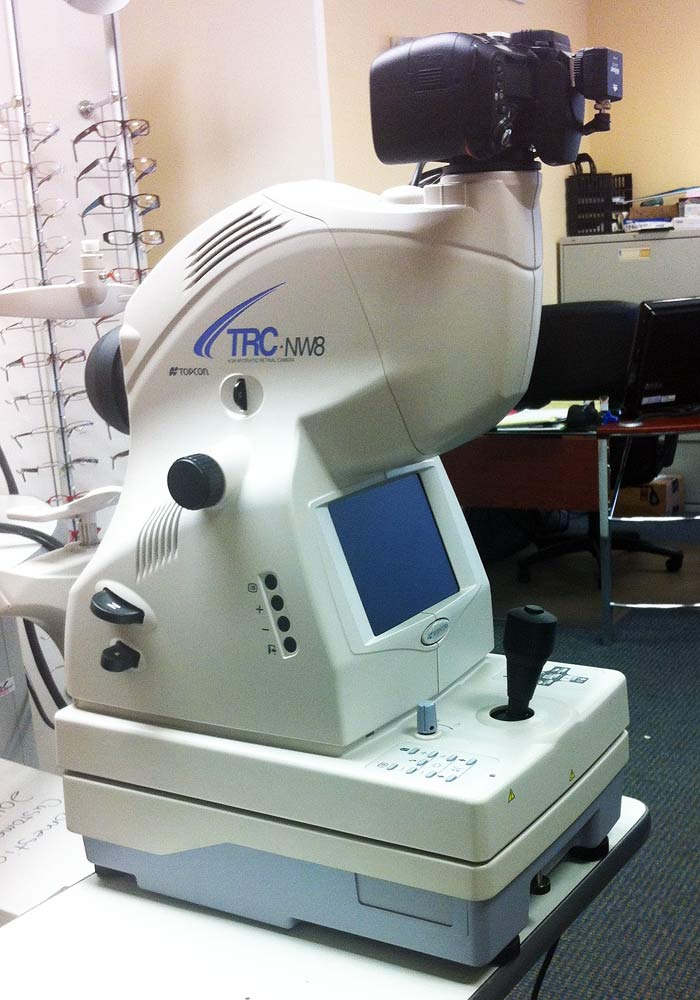
CASSI uses a coded aperture (mask) and a dispersive element to modulate the input scene, and it captures a two-dimensional (2D), multiplexed projection of a spectral datacube. Among these methods, only CASSI can measure a large-sized spectral datacube because it uses compressive sensing to acquire data 16, 17, leading to a high resolution along both spatial and spectral dimensions. In this category, representative techniques include computed tomographic imaging spectrometer (CTIS) 9, 10, the four-dimensional imaging spectrometer (4D-IS) 11, image mapping spectrometer (IMS) 12, 13 and coded aperture snapshot spectral imagers (CASSI) 14, 15. Additionally, it is challenging to keep a patient fixating on a target for an extended period of time.Ī snapshot spectral imaging system can avoid all these problems and provide an ideal solution for obtaining retinal spectral data. Particularly in retinal imaging, post-acquisition registration can result in artifacts due to tissue movement between successive images caused by arterial pulses as well as changes in the lens-eye geometry 9. Furthermore, the data acquired from sequential measurements need to be registered in post-processing, a complicated procedure that is sensitive to motion and image signal-to-noise ratio (SNR). The scanning mechanism typically leads to a prolonged acquisition, making these techniques prone to motion artifacts. To measure a spectral datacube (x, y, λ), conventional spectral imaging cameras rely on scanning, either in the spatial domain, such as using a slit scanning spectrometer 7, or in the spectral domain, such as using a liquid-crystal-tunable-filter 8. The overall rationale of using a spectral camera in retinal imaging is that the ocular tissue's endogenous optical properties, such as absorption and scattering, change during the progression of a retinal disease, and the spectrum of light emitted from tissue carries quantitative diagnostic information about tissue pathology. Originally developed for remote sensing 5, spectral imaging has gained increasing popularity in medical applications, including retinal imaging 6. The rich information could be used to classify the underlying components of the object. acquiring both spatial coordinates (x, y) and wavelengths (λ) of a scene simultaneously. 
In contrast, spectral imaging captures light in three dimensions, i.e. Despite being extensively used in clinics, these techniques measure only spatial information of the retina. The current standard-of-care retinal imaging technologies encompass color fundus photography, scanning laser ophthalmoscopes (SLO) 3, and optical coherence tomography (OCT) 4. 
Retinal imaging is crucial for the detection and management of ophthalmic diseases, such as age-related macular degeneration (AMD) 1 and glaucoma 2.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
